|
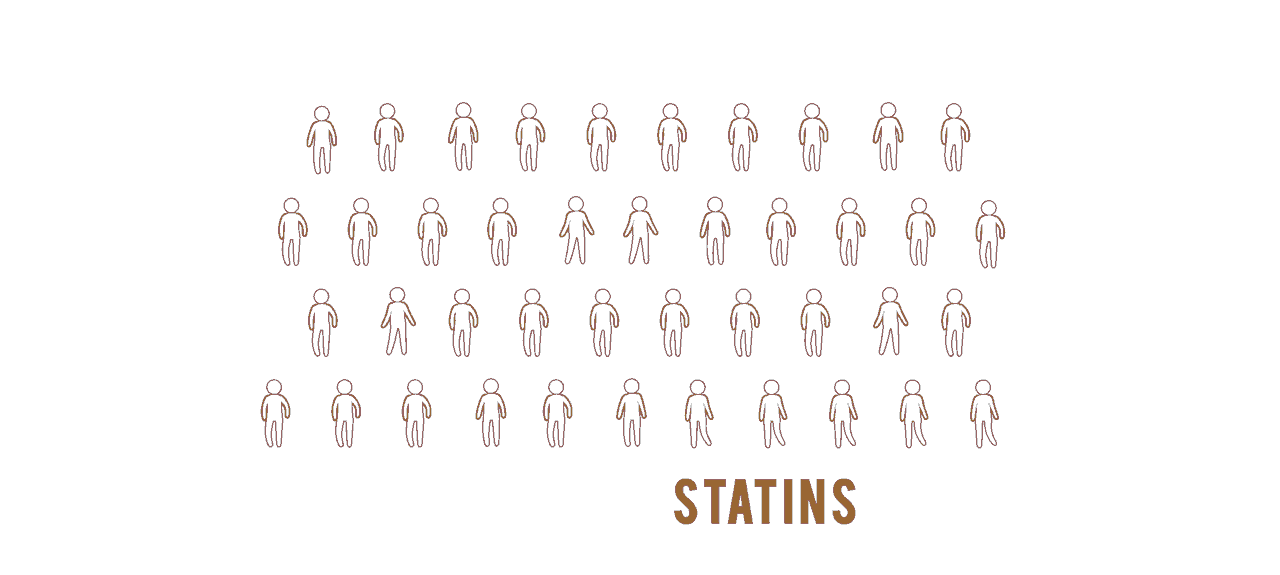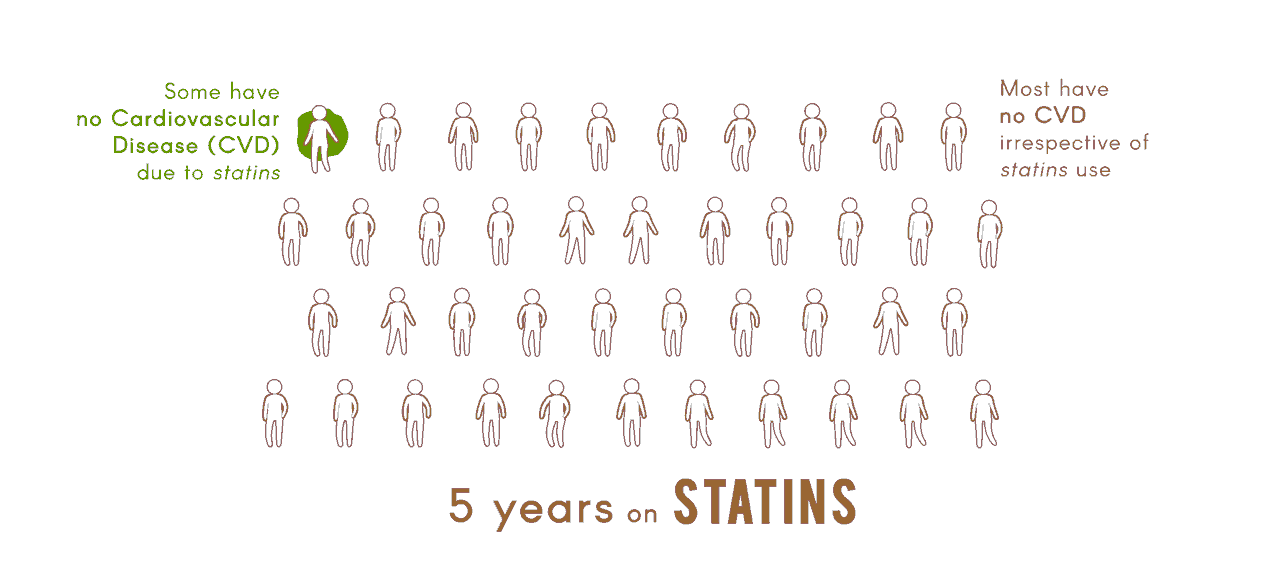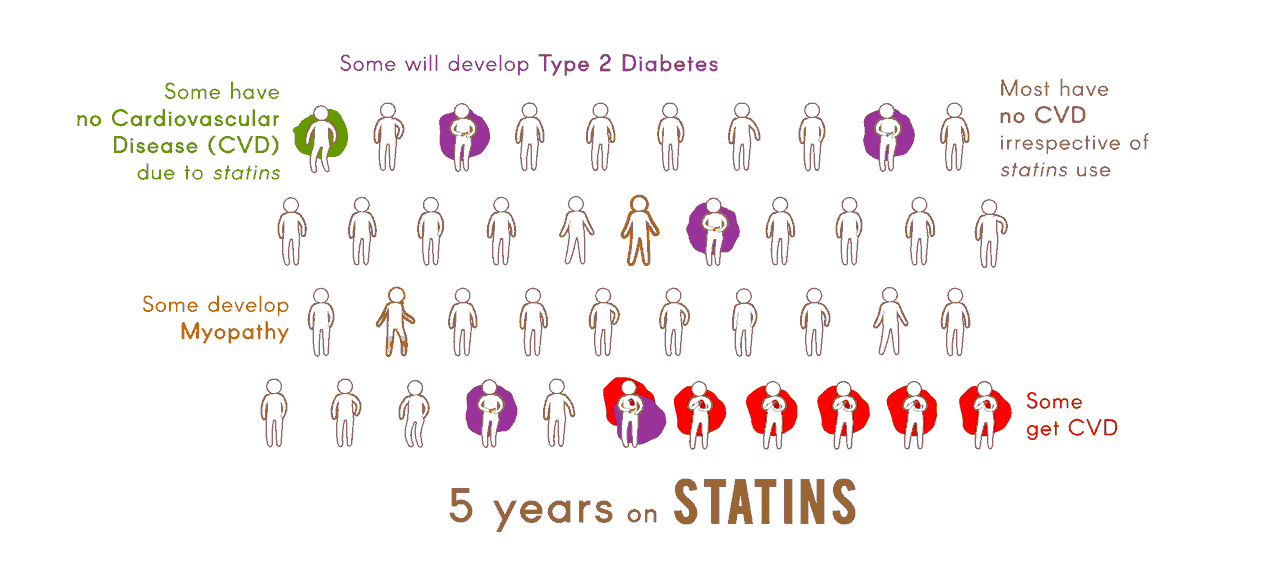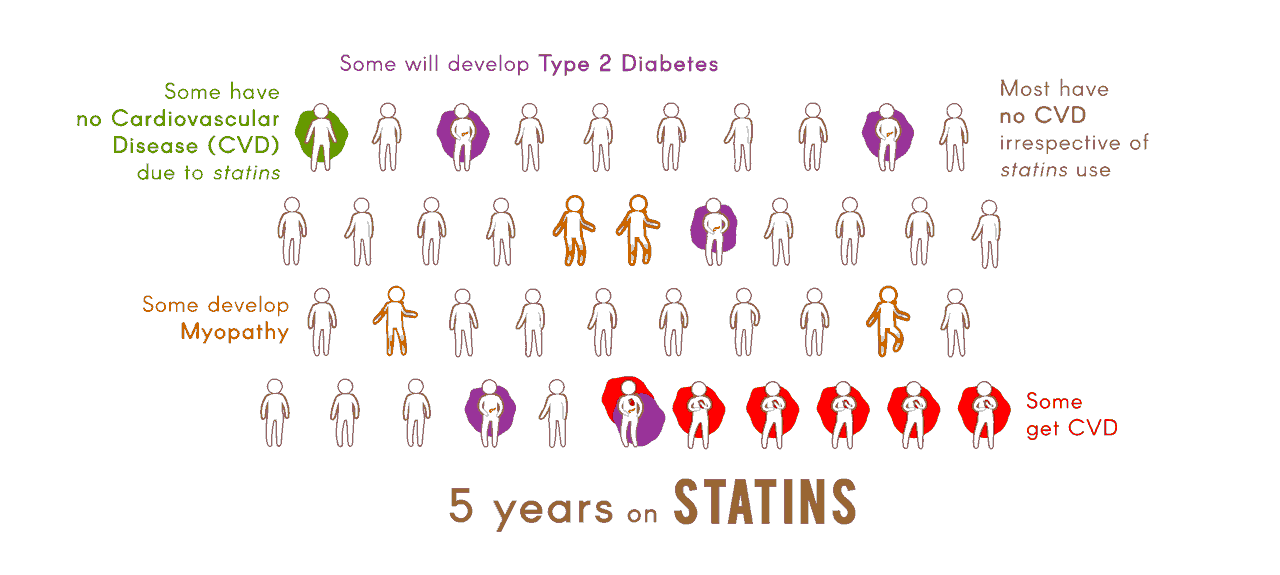
The overall objective of the Center "Pharmacogenomics of Statin Therapy" (POST) is to apply genomic, transcriptomic, and metabolomic analyses, together with studies in cellular and animal models, and innovative informatic tools, to identify and validate biomarkers for efficacy of statin drugs in reducing risk of cardiovascular disease (CVD), and for adverse effects of statins, specifically myopathy and type 2 diabetes.
The Center is comprised of three Projects, two Research Cores, and an Administrative Core. A key function of the Informatics Core is to integrate information obtained across the diverse yet complementary platforms of the three POST projects, thus fully leveraging each dataset to maximize understanding of the factors underlying variation in statin response. The Clinical Core at the Division of Research, Kaiser Permanente of Northern California serves a vital role by providing all the human participants for Project 1, and the genotypic data, phenotypic data, and DNA samples for Project 3. |
Molecular determinants of statin efficacy and adverse effectsA major aim of Project 1 is the identification of cellular transcriptomic and metabolomic markers for clinical efficacy and adverse effects of statins.
|
Identification of loci and validation of candidate molecular pathwaysProject 2 will use a unique, well-characterized panel of 100 inbred mouse strains to discover genetic variation associated with statin-induced myopathy and dysglycemia.
|
Genomewide association study of statin response and its consequenceIn Project 3, information will be leveraged to identify and replicate genetic associations with statin efficacy and adverse effects, as well as to assess the overall heritability of these responses.
|
|
Aim 1. Using existing genome-wide genotype data, and information from electronic health and pharmacy records from each of four race/ethnicity groups totaling over 46,000 statin users, identify common and rare genetic variants and environmental factors associated with statin clinical efficacy (reduction in lipids, MACE, and stroke), and side effects (myopathy and type 2 diabetes).
Aim 2. Estimate overall heritability of efficacy and adverse outcomes of Aim 1, and variation explained by identified variants. Aim 3. Perform additional genotyping using a custom designed exome array to replicate and extend findings. |
