Clinical application of pharmacogenomics knowledge will result in less ‘trial and error’ prescribing and more efficacious, safer and cost-effective drug therapy. However, despite the major advances in PGx and several commercially available PGx tests, its implication in routine patient care remains limited. The Ubiquitous Pharmacogenomics Consortium (U-PGx; www.upgx.eu) is a European project bringing together a large group of pharmacogenomics experts from institutes in 10 different countries. U-PGx entails a 5 year, 15 million euro programme funded by the European Union’s Horizon 2020 research and innovation programme. The ultimate objective of the consortium is to make actionable pharmacogenomics data and effective treatment optimization accessible to every European citizen. Specifically, U-PGx will investigate if the emerging approach of pre-emptive genotyping of an entire panel of important PGx markers will result in an improved outcome for patients and is cost-effective.
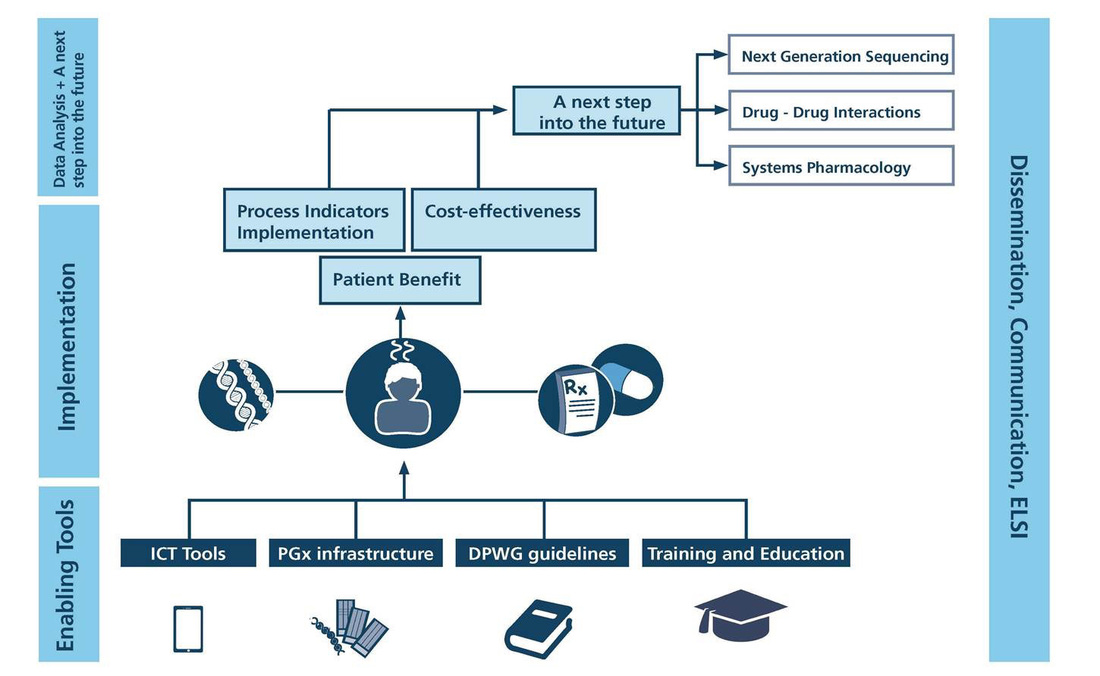
U-PGx uses a multifaceted approach consisting of four components to achieve this goal (fig 1). The first component focuses on developing enabling tools required to integrate PGx test results into the electronic healthcare record and computerized decision support systems, taking into account the differences in health care models, languages and laws across the EU. These enabling tools consist of IT solutions, PGx testing infrastructure, PGx educational programmes for healthcare professionals, and the translation of the Dutch PGx guidelines into local languages. This component will pave the way for the execution of component two. Component 2 entails implementation of PGx testing of a panel of pharmacogenes into clinical practice in a large cluster-randomised controlled trial (n=8,000) in 7 European countries(Netherlands, Spain, UK, Italy, Austria, Greece and Slovenia) , assessing the aggregate impact of multiple pharmacogenomics interventions on patient outcomes. Additional outcomes include, cost-effectiveness and process indicators for implementation and provider adoption of PGx. A third component applies innovative methodologies such as NGS and systems pharmacology to discover additional variants associated with drug response and to elucidate drug-drug-gene interactions. The final, fourth, component assures ethical proceeding of the project and spearheads outreaching and educational activities to influential stakeholders.