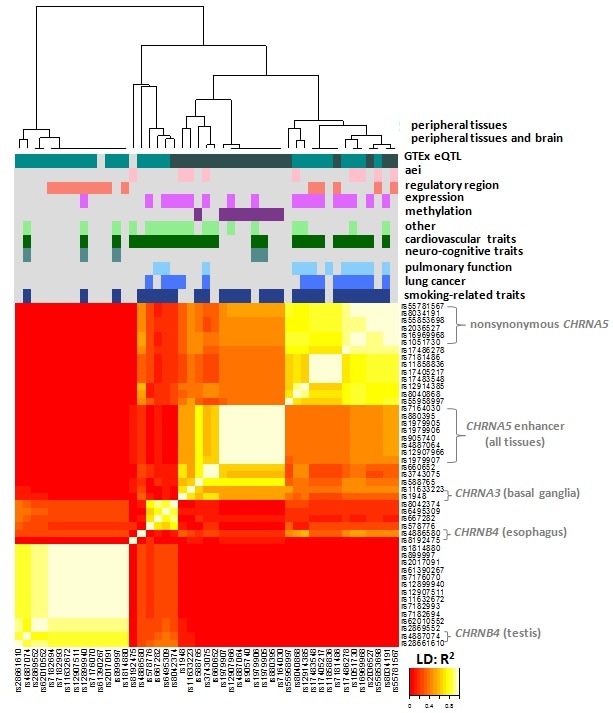
Introduction and Rationale. The vast majority of GWAS hits point to regulatory variants accounting for associations with complex traits, including drug response. Critical hub genes in biological networks tend to be under evolutionary selection, driving either purifying or positive selection. We have found that many drug target genes carry regulatory variants under positive selection, with high allele frequencies and influence on phenotypic traits. In most of these cases, more than one variant has arisen per gene locus in various populations. Finding the causative variants, and their interactions within the same gene locus, is critical for assessing the overall genetic influence, but this knowledge is still lacking for most genes. Even in heavy studied genes such as CYP3A4 and CYP2D6, we find substantial new regulatory variants that should be considered in biomarker panels (1,2). We posit that a full understanding of all functional variants and their interactions is key to evaluating the influence of each gene locus, and further, that dynamic interactions between gene loci (epistasis) cannot be fully revealed unless this overall influence is taken into account (statistically, a finding of epistasis is rapidly lost with decreasing LD of a surrogate marker with a causative SNP; ignoring the combined effect of multiple SNPs in a gene locus exacerbates this hurdle). As an example, we have selected the CHRNA5-CHRNA3-CHRNB4 nicotinic receptor gene cluster, containing protein coding and non-coding genes covered by long and frequent LD blocks, a signature of positive selection across the entire cluster (3).
Approach. We have develop an R package (K. Hartman) to canvass multiple databases including GTEx, dbGaP, ENCODE, 1,000 genomes project, etc, to extract LD, eQTLs, GWAS hits, and chromatin annotations, applicable to hundreds of genes at a time. This was applied to the nicotinic gene cluster to determine the main LD blocks and candidate variants associated with expression in body tissues and GWAS hits (3).
Results. In a first step, we have identified the main haplotype blocks and three SNPs, the regulatory variants rs880395 and rs1948, plus the known nonsynonymous CHRNA5 rs16969968, a known risk variant in nicotine dependence. Importantly, rs880395 affects the RNA expression of CHRNA3 and 5, and of an antisense RNA, while rs1948 affects specifically CHRNA3 only in the basal ganglia – a region important to nicotine dependence. The predominant LD structure in the gene cluster enables assignment of haplotypes and diplotypes with high confidence in a majority a study cohort. These results document the interactive nature of variants in this gene locus, modified in a tissue specific manner critical for determining influence on specific target traits.
Analysis of a GWAS cohort with nicotine dependence confirmed the known influence of the nsSNP alone, but haplotype and diplotype analyses reveal significant modulation of the influence of rs16969968 on nicotine dependence.
Conclusion. The CHRNA5-CHRNA3-CHRNB4 nicotinic receptor gene cluster represents a local regulome that should be considered for its overall influence on complex traits such as nicotine dependence. Operating via DNA looping over long distances, each regulatory variant can modify expression of multiple genes in an interactive fashion. We are now systematically expanding our approach to multiple gene loci and gene clusters, including cardiovascular and CNS disorder gene candidates, and genes in the innate immune system, which is under strong evolutionary constraints.
Approach. We have develop an R package (K. Hartman) to canvass multiple databases including GTEx, dbGaP, ENCODE, 1,000 genomes project, etc, to extract LD, eQTLs, GWAS hits, and chromatin annotations, applicable to hundreds of genes at a time. This was applied to the nicotinic gene cluster to determine the main LD blocks and candidate variants associated with expression in body tissues and GWAS hits (3).
Results. In a first step, we have identified the main haplotype blocks and three SNPs, the regulatory variants rs880395 and rs1948, plus the known nonsynonymous CHRNA5 rs16969968, a known risk variant in nicotine dependence. Importantly, rs880395 affects the RNA expression of CHRNA3 and 5, and of an antisense RNA, while rs1948 affects specifically CHRNA3 only in the basal ganglia – a region important to nicotine dependence. The predominant LD structure in the gene cluster enables assignment of haplotypes and diplotypes with high confidence in a majority a study cohort. These results document the interactive nature of variants in this gene locus, modified in a tissue specific manner critical for determining influence on specific target traits.
Analysis of a GWAS cohort with nicotine dependence confirmed the known influence of the nsSNP alone, but haplotype and diplotype analyses reveal significant modulation of the influence of rs16969968 on nicotine dependence.
Conclusion. The CHRNA5-CHRNA3-CHRNB4 nicotinic receptor gene cluster represents a local regulome that should be considered for its overall influence on complex traits such as nicotine dependence. Operating via DNA looping over long distances, each regulatory variant can modify expression of multiple genes in an interactive fashion. We are now systematically expanding our approach to multiple gene loci and gene clusters, including cardiovascular and CNS disorder gene candidates, and genes in the innate immune system, which is under strong evolutionary constraints.
- Intronic polymorphism in CYP3A4 affects hepatic expression and response to statin drugs. D. Wang, Y. Guo, S.A. Wrighton, G.E. Cooke, W. Sadee. Pharmacogenomics J 11: 274-286 (2011). PMID 20386561
- D. Wang, M.J. Poi, X. Sun, A. Gaedigk, J.S. Leeder, W. Sadee. Common CYP2D6 Polymorphisms Affecting Alternative Splicing and Transcription: Long-range Haplotypes with Two Regulatory Variants Modulate CYP2D6 Activity. Hum.Mol.Gen. 23: 268-278 (2014). PMID:23985325.
- E.S. Barrie, K. Hartmann, S.-H. Lee, J.T. Frater, M. Seweryn, D. Wang, W. Sadee. The CHRNA5/CHRNA3/CHRNB4 nicotinic receptor regulome: genomic architecture, regulatory variants, and clinical associations. Human Mutation, in print (2016). PMID:27758088, doi: 10.1002/humu.23135. https://www.ncbi.nlm.nih.gov/pubmed/27758088